PDF) Effect of Fimasartan versus Valsartan and Olmesartan on Office and Ambulatory Blood Pressure in Korean Patients with Mild-to-Moderate Essential Hypertension: A Randomized, Double-Blind, Active Control, Three-Parallel Group, Forced Titration ...
![PDF] UCLH GUIDELINES ON ANGIOTENSIN II RECEPTOR BLOCKERS (ARBs) FOR HYPERTENSION AND HEART FAILURE | Semantic Scholar PDF] UCLH GUIDELINES ON ANGIOTENSIN II RECEPTOR BLOCKERS (ARBs) FOR HYPERTENSION AND HEART FAILURE | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/74b2a0a4f852dbf2d9e67bb2ea87fda6cfd4a991/4-Table2-1.png)
PDF] UCLH GUIDELINES ON ANGIOTENSIN II RECEPTOR BLOCKERS (ARBs) FOR HYPERTENSION AND HEART FAILURE | Semantic Scholar
Angiotensin Converting-Enzyme Inhibitors, Angiotensin Receptor Blockers, and Calcium Channel Blockers Are Associated with Prolonged Vascular Access Patency in Uremic Patients Undergoing Hemodialysis
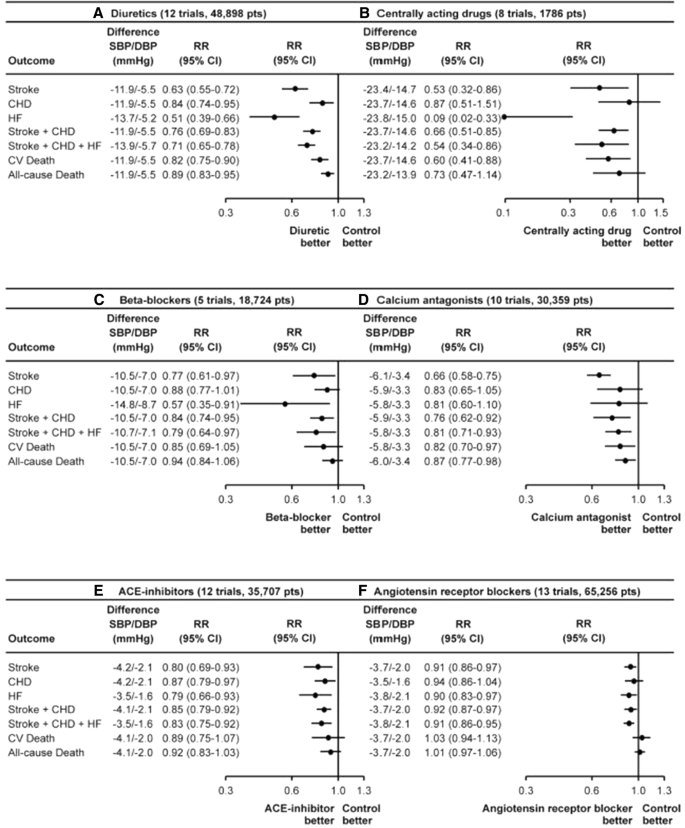
Angiotensin Receptor Blockers Versus Angiotensin Converting Enzyme Inhibitors for the Treatment of Arterial Hypertension and the Role of Olmesartan | SpringerLink
FDA Advise-ERR: Concomitant use of Entresto and ACE inhibitors Can Lead to Serious Outcomes | Institute For Safe Medication Practices
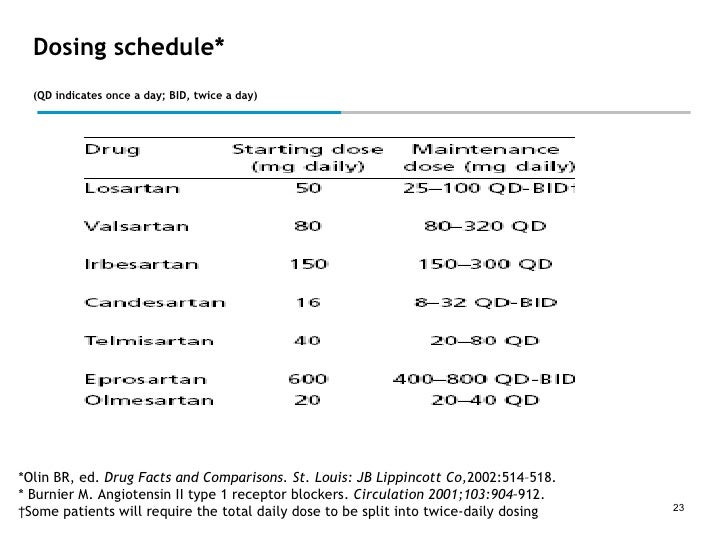
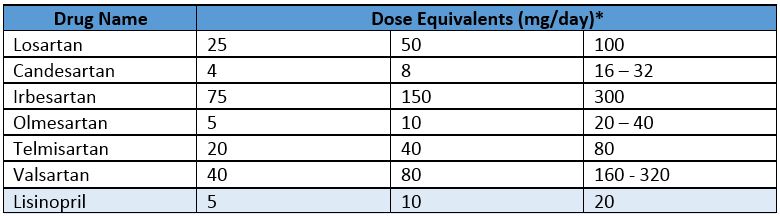
Equivalent dosing of irbesartan, valsartan, and losartan identified through formulary switch at a Veterans Affairs medical center
![PDF] Olmesartan Reducing Incidence of Endstage Renal Disease in Diabetic Nephropathy Trial (ORIENT): Rationale and Study Design | Semantic Scholar PDF] Olmesartan Reducing Incidence of Endstage Renal Disease in Diabetic Nephropathy Trial (ORIENT): Rationale and Study Design | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bcf5a751b2ba4467266d3ebdddc74f2fd8ee0b51/5-Table3-1.png)